Drug discovery and development
We have created a pipeline of competitive oligonucleotide drug candidates – both proprietary and partnered – in the fields of (immuno)oncology, inflammatory, fibrotic or cardiometabolic diseases as well as diseases of the central nervous system (CNS) and neuromuscular diseases. Our candidates are in various stages of pre-clinical and clinical development.
Our development efforts focus on targets, where our oligonucleotide therapeutics have the potential to be first-in-class or best-in-class, offering new hope to patients facing conditions that are beyond the reach of current approaches and modalities. For our proprietary lead program SECN-15, targeting Neuropilin 1 in the context of immuno-oncology, IND-enabling studies are underway to advance SECN-15 into clinical development. We continue to selectively broaden our pipeline with promising targets in areas of high unmet medical need.
Our Pipeline

SECN-15:
Unraveling the potential of oligonucleotide innovation in immuno-oncology
Our lead program SECN-15 is a potential first-in-class antisense oligonucleotide (ASO) targeting Neuropilin 1 (NRP1).
NRP1 is a protein which is involved in various tumor-promoting functions such as angiogenesis, suppression of effector immune responses, cancer cell migration and metastasis. In particular, NRP1 contributes to the suppressive capacity of tumor-associated macrophages and regulatory T cells, both cell types that can be efficiently targeted with ASOs.
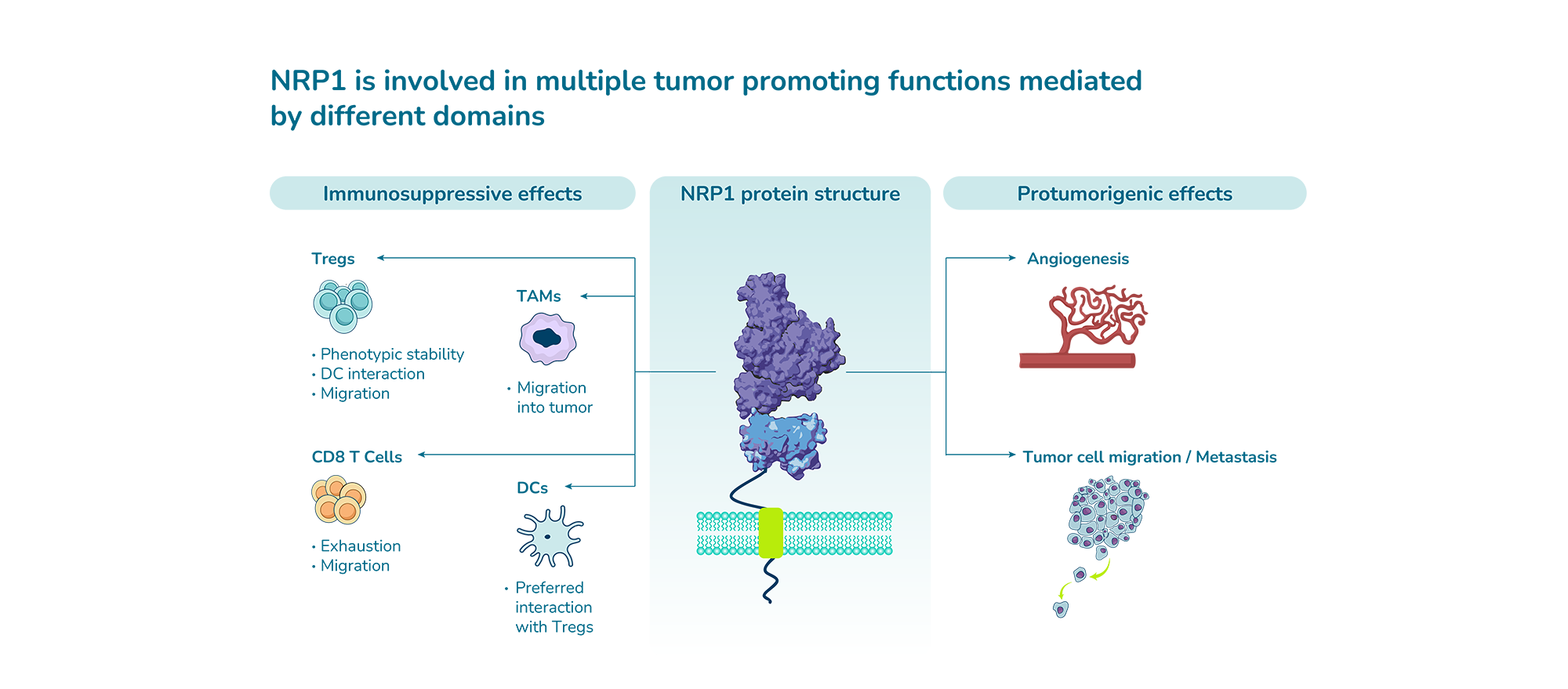
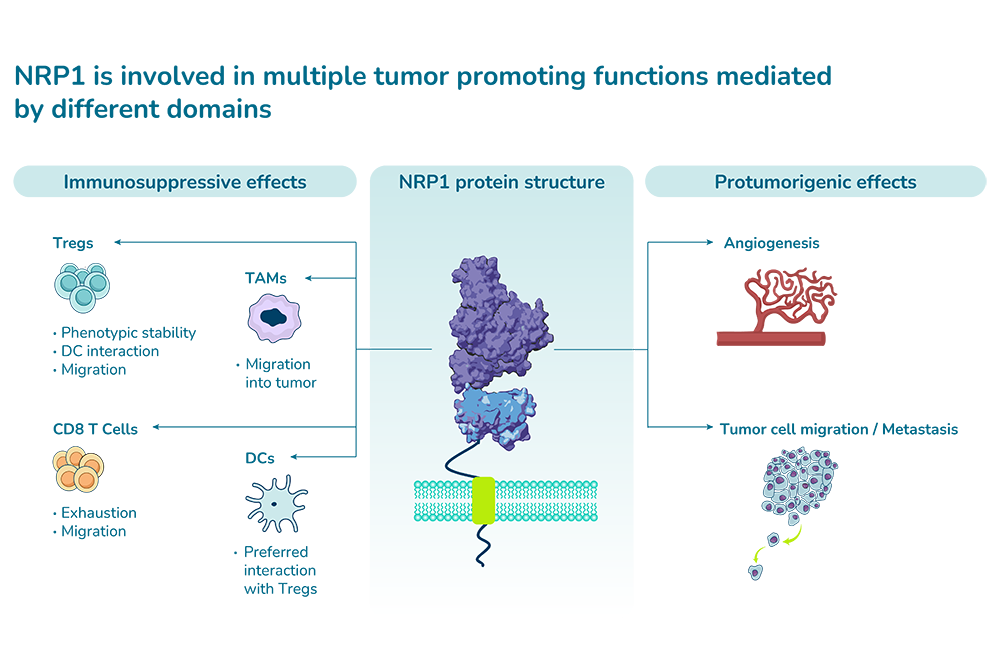
Due to pharmacokinetic hurdles posed by its soluble and endothelium-bound forms, NRP1 is a challenging target for approaches that bind directly to the target protein such as antibodies, peptides or small molecules. These hurdles do not apply to modalities that don’t bind to the target protein such as antisense oligonucleotides. Furthermore, different functions of NRP1 are mediated by different domains that cannot be blocked simultaneously by modalities that bind to the target protein; however, unlike these modalities, oligonucleotides prevent protein production by targeting the NRP1 RNA and therefore are able to simultaneously block all functions of NRP1.
SECN-15 selectively and with high potency suppresses NRP1 protein production in relevant immune cell types of the tumor microenvironment, translating into highly potent single agent and synergistic anti-tumor activity in combination with PD1/PD-L1 checkpoint inhibitors in in vivo disease models, including PD-1 checkpoint inhibitor resistant models.
SECN-15 has the potential to break resistance to checkpoint therapy in cancer patients and is promising in a range of solid tumors. Translational data confirm gastric cancer, a tumor type in which high NRP1 expression is linked to inferior outcomes, as potential lead indication.
SECN-15 clinical development plan
Our ongoing efforts involve advancing IND-enabling studies, positioning us for an IND submission in 2026.